Publicly available data suggests that a crime of negligence or malfeasance has been committed, and it’s possible it contributed to the deaths of patients in a UK clinical trial. The credit for this story must go to Xavier Azalbert of France Soir who did a huge amount of work on it. I’m writing it here with his permission because, to my knowledge, I don’t think the story has had anywhere near the coverage it deserves.
When patients across the UK fell seriously ill and became hospitalised with Covid-19, some were offered the chance to take part in a “RECOVERY” medical trial. Its primary investigators, Professor Martin Landray and Professor Peter Horby, had big money backing for RECOVERY to find a treatment for the raging pandemic. With few options available, family members of desperately ill Covid-19 patients would sign consent to the trial, but on the first day of treatment, the trial would administer an unusually large dose of hydroxychloroquine (HCQ) to those patients. This is the story of how such a thing happened, and what it might tell us about the pandemic response.
The background
When the Recovery Trial was announced in March 2020, it was supposed to investigate hydroxychloroquine as a potential treatment for Covid-19. Much like Ivermectin, hydroxychloroquine became a widely used but controversial drug during the pandemic. Much like Ivermectin, it’s a World Health Organisation “essential medicine” in use for decades, meaning its dose and toxicity have been well established. As I previously reported, chloroquine had been identified as a treatment for the original SARS virus all the way back in 2006.
That hydroxychloroquine might be effective against SARS-CoV-2 was hardly a radical idea. Just four weeks after SARS-CoV-2 had been formerly identified and long before most were even aware of a brewing pandemic, a research team from China had already submitted a study to Nature showing “Hydroxychloroquine can efficiently inhibit SARS-CoV-2 infection in vitro.” Four weeks later, a study in France was ready and registered, and by the 18th of March 2020, they had results that looked very promising. Led by Professor Didier Raoult, they showed that as time went on, Covid-19 patients taking a hydroxychloroquine therapy were recovering significantly faster, indicated by the green line below.

With the world growing increasingly anxious about rising cases, you would be tempted to think these results would be well received, but the investigators behind the trial were publicly mocked and derided in a series of media briefings. As I previously reported, many of the academics attacking Didier Raoult were in receipt of money from the pharmaceutical industry. Some were even candid enough to promote their own trials as they talked down these positive results. What was going on? The attacks took the investigators by surprise and it marked the beginning of a series of incongruous developments in the pandemic response.
The attacks didn’t stop other researchers. All over the world, dozens of studies were rolling in that showed hydroxychloroquine was effective. If you’re familiar with the rest of my work, you’ll understand the pattern by now; the next stage is for the “correct” people and institutions to investigate the drug and study it “properly.” Those “proper” institutions then present an “unambiguous and clear answer” on the drug’s usefulness.
Forget the Didier Raoult study, the independent researchers and the fact that entire nations had already adopted the drug for use because The RECOVERY trial was here, and it was going to answer the question definitively. It’s worth pointing out, the trial was funded by the Wellcome Trust and the Bill and Melinda Gates Foundation. The editorial of these moments always sits silently between the lines; the competent people are here! Sitting in their plush and industry-funded lobby, they were going to tell us if Hydroxychloroquine worked or not. The lead investigators were Professor Peter Horby and Professor Martin Landray.
Professor Peter Horby announcing the RECOVERY trial in early 2020
Despite the hyper-competent veneer, there was an inexplicable decision resting at the heart of the RECOVERY trial, one which ought to have been challenged by journalists if we didn’t live in a sermon/congregation paradigm of “The Science”. What was this strange issue?
For those on the front line treating Covid patients, it was clear that combination therapy would be the way to go. That means combining multiple drugs and interventions together to create a synergistic effect. It’s why Didier Raoult’s team used hydroxychloroquine and azithromycin together, and you can clearly see the difference it made in Figure 1 above. Why then, did the RECOVERY trial insist on investigating each drug entirely in isolation? It is a confusing riddle. Was it possible the trial wasn’t really looking for effective treatments, it was instead a tool to clear a pathway for novel pharmaceuticals to hit the market? We can only speculate. Needless to say, by April 3rd 2020, the RECOVERY trial announced it would investigate hydroxychloroquine as a monotherapy treatment for Covid-19. It was immediately following this announcement that things start to get strange.
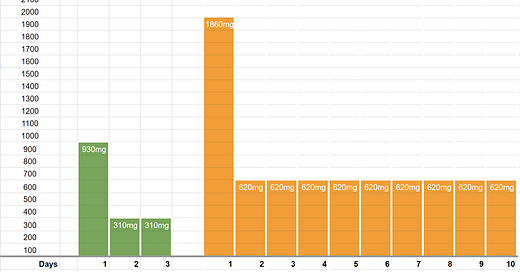
When researchers put a trial together, they register the protocol publicly. It tells other researchers what the trial plans to do and how. It normally contains a lot of technical detail on how the investigators plan to run their study. The first mention of hydroxychloroquine was in protocol version 3 on April 7th 2020. The investigators state the dose they plan to give to patients; an initial dose of 1840mg within the first 24 hours.

1860mg is a very high dose. It’s one that would require careful justification, and it’s a dose we’re now going to explore. So how was such a dose justified? A few paragraphs before the section I’ve quoted above in Figure 2, the investigators reference the HCQ dose used in the treatment of acute malaria in order to support their proposed dose for Covid-19. To be clear, I’ll quote exactly what they said:
Initially 620 mg,
then 310 mg after 6-8 hours,
then 310 mg daily for 2 days.
This is equivalent to 930mg base in the first 24 hours.
So the dose over 24 hours for acute malaria is 930mg, and Landray and Horby say they are doubling this to 1860mg in the treatment of Covid-19. For some reason, they don’t directly reference this malaria guideline, I had to look for it, and I believe the one they’re referring to is this UK guideline on treating malaria.
Because the guideline they’re relying on is to treat acute malaria, the doses it suggests are already high. Acute malaria is no joke. As such, the guideline issued warnings about the risks of these suggested doses, “If intravenous quinine needs to be continued for longer than 48 h, or the patient is in renal failure or has severe hepatic dysfunction, quinine doses should be reduced by a third.” To translate, at the doses the guideline suggests, doctors might see kidney failure or liver damage and they must dial down their doses accordingly. These same malaria guidelines go on to say “caution should be exercised in older patients or those with cardiac disease, because of the potential for quinine to lead to arrhythmias. These patients should have ECG monitoring during intravenous quinine treatment.” To translate, at these doses some patients will need need to have their hearts monitored because it can lead to heart irregularities, which can be fatal.
Right there, in the very guidelines they’re referencing, there are warnings that the doses are already high enough to cause serious side effects. So, with that in mind, what justification might there be for doubling a dose that is already bordering on toxic? Landray and Horby say “the objective is to reach plasma concentrations that are inhibitory to the virus as soon as safely possible. Given the significant mortality in patients hospitalised with COVID-19 this dose is felt to be justified.”
They felt it was justified.

But is this really just a double dose as the investigators have claimed? The dosing schedule they used works out to 8060mg base of HCQ because their dosing schedule is actually 10 days long. The malaria guideline suggests a total recommended dose, saying “five to seven days of quinine in total”, later it says “five days treatment is adequate for the vast majority of cases”
So not only are Landray and Horby doubling the size of each individual dose, they’re doubling the length of the treatment as well. This is another way to increase the dose, and this really matters because drugs build up in our body’s blood. One could even argue that they’re more than doubling the length of the treatment, because if we look carefully at the guidelines they’re using to justify all of this, in the part about the specific type of malaria they mentioned, it says to use “25 mg/kg in total over 3 days”. For an average-sized person, that would be a total of 1240mg over 3 days, but Landray and Horby planned to use a total dose of 8060mg over 10 days. They’re suggesting 6.5x the total dose of the suggested dose to treat malaria.
Looking at them side by side like this, it’s clear there’s a huge discrepancy in the dose. Had Landray and Horby even considered that their dose was potentially toxic? They were administering a drug well in excess of the UK guidelines, so we might reasonably assume they searched the medical literature for chloroquine and hydroxychloroquine toxicity. So I followed in what I presumed would be their footprints to find out at what dose hydroxychloroquine becomes toxic. Documenting a toxic dose of a drug via experiment is obviously very difficult because you basically have to poison someone to do that. Because of this obvious barrier, individual case studies on drug toxicity become very important. Occasionally, people ingest too much of a drug by mistake, or sometimes in suicide attempts. As grisly as these cases are, they become invaluable in understanding toxicity.
In this case study, published in 2017, a lady ingested 3000mg* of hydroxychloroquine by mistake. It had fallen into her cup of coffee whilst in the car and she drank the coffee. Based on that, investigators estimated she had ingested 3000mg*. It messed her up pretty bad: tinnitus, facial and lower extremity paresthesias, abnormalities in her peripheral vision, postural disequilibrium, neurotoxic vestibulopathy, chronic ataxia, and visual disturbance. Twelve weeks after the dose she made only mild improvement. By any objective measure, this was a poisonous dose.
In that same paper, authors say that “the consumption of more than 4000mg* of hydroxychloroquine is generally accepted as a severe intoxication based on the onset fatal ventricular arrhythmias”. Put simply, 4000mg* of HCQ is a potentially life-threatening dose and the authors of that case study clearly showed that 3000mg* certainly risks poisoning patients quite badly. It’s worth pointing out here that the authors are talking about the tablet dose, not the base dose, which is why the numbers are slightly different. But comparing like for like, the RECOVERY trial opted to give 2400mg of HCQ, followed by 800mg every 24 hours for ten days, even though this case study shows that a 3000mg dose is very likely to be toxic. Horby and Landray’s total cumulative dose is a total of 10.4 grams of hydroxychloroquine. This cumulative build-up of HCQ really matters, so assuming this case study is accurate, the investigators are using a dose that's very likely to be toxic. Let me explain why.
* With HCQ dosing, there’s an mg dose, which is the size of the tablet, and a “base” dose, which is the quantity of active substance in the tablet. We’ve been talking about “base” dose, because that’s what all the guidelines use and it’s what Landray and Horby use. In these case studies, they’re talking about mg dose, which is slightly larger. Horby and Landray’s proposed ‘loading dose’ was 2400mg, which works out to a base dose of 1860mg.
When you’re given drugs in multiple doses, the doctors have to consider how rapidly or slowly your body can clear the drug from your blood. That way they can dose you in a ‘steady state’. A known way to measure a drug’s clearance from the body is through a drug’s half-life. How long does it take your body to clear out half of the drug from your blood? In the case of HQC, the half-life is actually quite long at 22.4 days. This is a known fact about HCQ, and it’s stated clearly by the FDA itself.
It means that the schedule Landray and Horby used ensured that HCQ is building up in their patient’s blood. To illustrate this, I’ve used a crude drug half-life calculator from grams.io. It allows us to compare the drug levels we might expect to see building up in a patient’s blood as per Landray and Horby’s proposed dosing schedule. We can compare that with a known toxic dose as documented by the case study we just looked at. The line in purple is the toxic dose from the case study above, the line in green is the dose proposed by Landray and Horby in the RECOVERY trial.

As you can see above, around 41 hours after their dosing starts the patients arguably have a known toxic quantity of HCQ in their blood, but as you can see it gets worse because they keep dosing for an additional eight days after crossing this threshold and reach a maximum active dose of 8 grams.
So maybe the case study is wrong? Because it seems too bizarre that investigators took the guideline dose to treat acute malaria, doubled the individual doses, doubled the length of treatment, and then gave that dose to patients? They must have checked if this unusual dose carried toxic risks?
But again, I was surprised to find that the statements on maximum doses are clear. On the FDA drug label for hydroxychloroquine, it states clearly that even in the treatment of malaria, the dose is “not to exceed 620 mg base”, and yet Landray and Horby use twice this dose. After the initial loading dose, the FDA says “not to exceed 310 mg base”, and again, Horby and Landray used twice this dose. How can we justify doubling a dose that the FDA specifically say should not be exceeded? Based on FDA guidelines, a threshold was crossed that shouldn’t have been.
There’s also the UK database on toxic substances. Look at hydroxychloroquine and you’ll get a clear answer, “Adults who have ingested 10 mg/kg or more chloroquine base as a single dose should be referred for medical assessment“. The initial dose that patient’s received on the RECOVERY trial was 620mg, which means that anyone weighing 62kg or less received a toxic dose from the first four tablets on Landray and Horby’s dosing schedule. As you can see from Figure 4 above, even though those doses were split into two 12-hour intervals, those patients received 620mg every single day for 10 days. The average weight for a woman in the UK is 70kg, which means that it’s very likely there were women in the study who weighed 62kg, and therefore, according to very clear information on toxbase, those patients recieved toxic doses.
Surely the investigators wouldn’t have been so careless as to not adjust doses based on a patient’s weight? But right up until May 2020, the protocol clearly says on page 23, “No dose adjustment is required for weight based on the doses defined in this protocol”. The people weighing 62kg or less on the trial received toxic doses on their first day of the trial. Based on the cumulative doses they then administered, totalling 620mg every day regardless of the patient’s weight, it’s very plausible that other patients on the trial were poisoned too. How can such a mistake have been made?
Noticing the dose seemed very large, we’re lucky to have the work of Xavier Azalbert of France Soir, upon which this article is based. His publication had the chance to interview Professor Martin Landray on June 5 at 8:00 p.m. Suspecting a problem, Azalbert asked Professor Landray about it directly, and Landray’s answer creates more questions than it answers.
France Soir: Could you specify the dosage of HCQ that you gave to the patients?
Martin Landray: It is 2400mg* in the first 24 hours and 800mg* from day 2 to day 10. It is a treatment for 10 days in total. These are high enough doses to ensure that the level of HCQ in the blood is high enough to have a chance of killing the virus
France Soir: How did you decide on the HCQ dosage?
Martin Landray: The doses were chosen based on pharmacokinetic modeling and these are in line with dosages used for other diseases such as amoebic dysentery .
France Soir: Is there a maximum dosage for hydroxychloroquine in the UK?
Martin Landray: I have to check, but it's much bigger than 2400mg, I think it's six or ten times more. For Covid, there is no recommended dose because it is a new disease
Audio from the exchange extracted from the interview Landray did with France Soir. It’s available at this link.
This is an incredible exchange. It suggests something very strange was going on with the dosing on the hydroxychloroquine trial. He’s arguing the dose is based on the treatment for amoebic dysentery, even though his own protocol says the dose is based on the treatment of acute malaria. To make matters stranger, amoebic dysentery isn’t usually treated with hydroxychloroquine, a drug called Metronidazole is used. That’s according to the UK’s NICE guidelines.
As we saw from the official database of UK poisons, a dose of 10mg per kilo of weight means a patient should seek medical attention. The 10x dose Landray suggests would be fine works out to 24,000mg, or for the average woman, a dose of 342mg per kilo. This dose is 34x more than what is considered a poisonous dose. It is unambiguously an overdose, which even a quick glance through the medical literature confirms.
Either Landray is lying, or more likely, he’s catastrophically mistaken. The question of critical importance is how he became so mistaken over such a critical component of the trial. An open investigation, revealing all of the trial’s data, is the only way this could be properly understood. This mistake, if it is a mistake, benefitted the pharmaceutical industry because it could have hidden a signal of benefit and in the process created a valuable tool to bash a cheap and highly available drug. A drug that would have taken dollars away from the novel pharmaceutical drugs which were well on their way to market at this time. As grisly as it may be, a medical trial that poisons people could be beneficial to a pharmaceutical industry looking to sell novel drugs. Sometimes market forces can force a profitable mistake…
In the interest of not creating articles that stretch to breaking point, I’m breaking this series of articles up and will release more soon.








Excellent article as always Phil. However in looking at this issue we need to look at the wider context of what was going on in the UK at the same time.
There were two types of trials ongoing in the UK at this time the Recovery Trial and Principle Trial. The Principle Trial looked at early treatment before hospitalisation.
The Principle Trial had an initial criteria of 7 days from onset of symptoms/test result to be admitted to the trial. This was strangely changed to 14 days for the Hydroxychloroquine trial. This alone raises a question about whether the trial was designed to fail.
Then we have the following timeline:
22 May 2020 The Lancet publishes study on Hydroxychloroquine, reports that hydroxychloroquine and chloroquine increased the risk of in-hospital death by 30% to 40% and increased arrhythmia by a factor of 2 to 5.
22 May 2020, The Medicines and Healthcare products Regulatory Authority (MHRA) instructed the PRINCIPLE trial of community-based treatments for COVID-19 illness to suspend recruitment into its hydroxychloroquine arm, based on the findings of an observational study of hospitalised patients in the Lancet.
24 May 2020 statement from the Recovery Trial ‘On Friday 22nd May we received a letter from the MHRA in which they notified us of their concerns relating to the use of hydroxychloroquine as a treatment for patients with COVID-19 in the light of the recent publication by Mehra et al in The Lancet on 22 May 2020.
We have held two videoconferences with the MHRA and provided a detailed response which is summarised below.
This morning we have received written confirmation from the MHRA that, “it is acceptable to allow continued randomisation into the hydroxychloroquine arm of the trial.”’
4 June 2020 The medical journal The Lancet on Thursday retracted the large study on the use of hydroxychloroquine to treat COVID-19 because of potential flaws in the research data.
So we have the Principle trial which was using the much smaller dosage 200mg per day, stopped straight away but the Recovery trial using the very dangerous dosage allowed to continue. Then we have Chris Butlers, lead investigator, comments that he was surprised that the Principle trial was stopped.
I hope you’re going to cover this in future articles because the context tells you a lot about what was going on. The whole thing stinks to me.
Just like ivermectin, but in reverse . IVM was generally way underdosed , not used long enough , and patients started too late in disease process . They would have had problems trying to “justify “ overdosing with it because it’s so safe . Negative results was the predetermined end point for $UCE$$ . And Gates achieved it across the board.